C2 2- Diamagnetic Or Paramagnetic
For this we volition first at the atomic orbitals and construct a molecular orbital (MO) diagram to be certain.
We discover that since
So and so, you lot're xc% of the way there. Since paramagnetism requires an unpaired electron, is
- How many more electrons does
#"C"_2^(-)# accept than#"C"_2# ? - Where does it go?
- Is it unpaired?
My arroyo begins like this:
- Carbon has access to its one
#\mathbf(1s)# , one#\mathbf(2s)# , and three#\mathbf(2p)# orbitals (with the#1s# orbital much lower in energy than the#2s# and#2p# 's). We don't have to care near the#1s# electrons; they can exist omitted from the MO diagram because they're so depression in energy. - Each carbon has four valence electrons: two occupy the same
#2s# orbital, and two singly occupy two of the three#2p# orbitals. - Since ane carbon has four valence electrons, two carbons bonded together must have a total of viii. This gives the number of valence electrons in
#"C"_2# , just non#"C"_2^(-)# . - The
#1s# orbital of each carbon combine caput-on to form a#\mathbf(sigma_"1s")# bonding and#\mathbf(sigma_"1s"^"*")# antibonding molecular orbital. - The
#2s# orbital of each carbon combine head-on to form a#\mathbf(sigma_"2s")# bonding and#\mathbf(sigma_"2s"^"*")# antibonding molecular orbital. - The
#2p_x# orbital of each carbon combine sidelong to course a#\mathbf(pi_(2p_x))# bonding and#\mathbf(pi_(2p_x)^"*")# antibonding molecular orbital. - The
#2p_y# orbital of each carbon combine sidelong to form a#\mathbf(pi_(2p_y))# bonding and#\mathbf(pi_(2p_y)^"*")# antibonding molecular orbital. - The
#2p_z# orbital of each carbon combine head-on to form a#\mathbf(sigma_(2p_z))# bonding and#\mathbf(sigma_(2p_z)^"*")# antibonding molecular orbital.
For
For
But... for
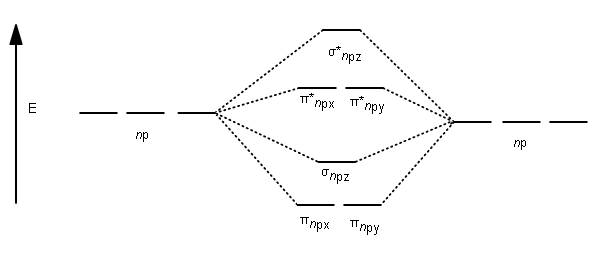
Therefore, combine steps 4-8 to accomplish the MO diagram for
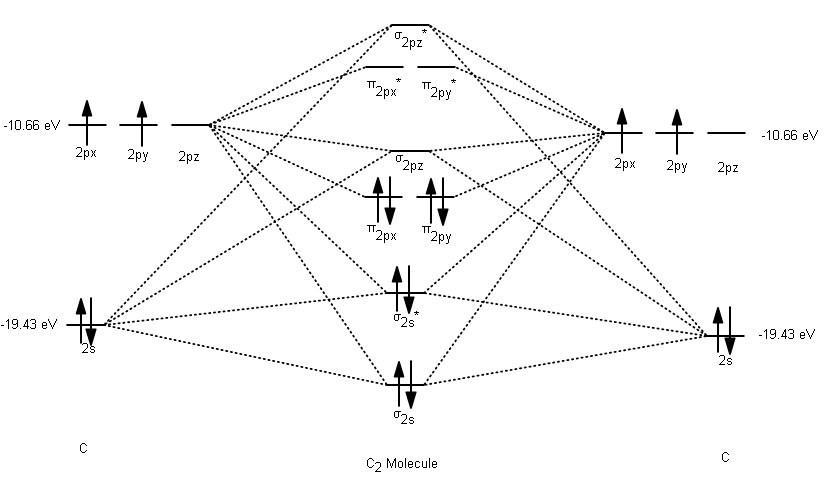
and
#(sigma_(1s))^two(sigma_(1s)^"*")^2stackrel("valence electrons")overbrace((sigma_(2s))^2(sigma_(2s)^"*")^ii(pi_(2p_x))^2(pi_(2p_y))^2)#
Since
So then, since paramagnetism requires an unpaired electron, is
C2 2- Diamagnetic Or Paramagnetic,
Source: https://socratic.org/questions/is-c-2-paramagnetic-or-diamagnetic-1
Posted by: holmesswuzzy1946.blogspot.com


0 Response to "C2 2- Diamagnetic Or Paramagnetic"
Post a Comment